
Research Projects
At the Center for Synthetic Immunity, we aim to develop novel cell therapeutics and move them into the clinic.
Synthetic Immunity is the intentional design and construction of new biological pathways, molecules and immunological systems to provide a means to induce, improve, or regulate specific disease pathways. Our research is focused on the use of chimeric antigen receptor (CAR) based cell therapy. CARs are used to target immune cells to specific disease associated antigens.
A disease may be a good candidate for CAR cell therapy if it is –
- Chronic, debilitating, or a fatal condition
- A significant health issue or threat
- A ‘complex’ pathological process where multiple factors contribute to the disease process
- Controlled by pro – or anti-inflammatory processes
We build designer immune cells that –
- Target key effector cells and mechanisms
- Alter the local microenvironment
- Repair the tissue
- Prevent further tissue damage
The successful development of novel CAR based cell therapeutics has the potential to treat many chronics and difficult to treat illnesses where there is a high unmet medical need.
NK cell receptor-based CAR therapy
CAR effector cells can be developed from T cells, NK cells, and macrophages. These CAR cells can target various types of cancer. CARs are designed to recognize specific target molecules on tumor cells, resulting in the killing of tumor cells and the activation of immune cells. This CAR activation leads to the direct killing of tumor cells and fundamentally transforms the tumor microenvironment, inducing an anti-tumor immune response and restricting tumor growth. We have developed a series of innovative CAR approaches based on natural killer cell receptors, including NKG2D, NKp30, and B7H6-specific CARs.
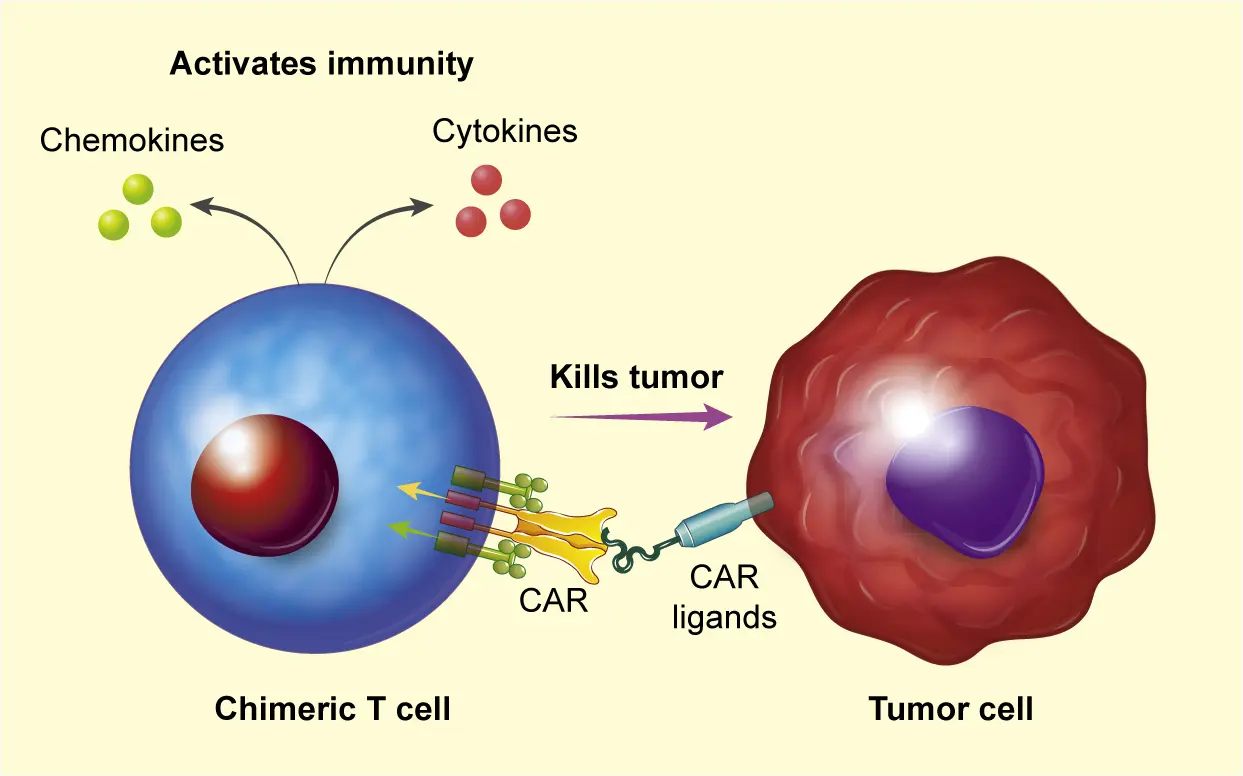

Allogeneic cell therapy
Our strategy employs healthy donor T cells as the foundation for CAR T cells. Not all patients have robust T cells suitable for autologous CAR T cell therapy. The advancement of technologies that allow the use of healthy donor T cells while preventing potential graft-versus-host disease (GVHD) would significantly improve the production and cost-effectiveness of CAR T cell therapy. We have developed TIM (T cell inhibitory molecules) technology to enable the use of healthy donor-derived CAR T cells without causing GVHD, while maintaining high therapeutic efficacy.
CAR Treg based therapy
Numerous diseases are characterized by an excessive inflammatory response, which can result in tissue pathology and the destruction of healthy tissues. To address this, we have engineered CARs to function within T regulatory cells (Tregs). These Tregs possess the capacity to modulate inflammation and restore homeostasis. Ensuring the purity and stability of Tregs is critical, as these factors are essential for maintaining their function and persistence in vivo. While our primary focus is on employing CAR Tregs for neurodegenerative diseases, their application extends to tissue transplantation, autoimmune disorders, and other chronic inflammatory conditions.

CAR T cell platforms
We are creating innovative platforms to boost the effectiveness of CAR T cells. These cell engineering platforms focus on enhancing efficacy, improving safety, and increasing persistence of CAR T cells in vivo. Our platform technologies can be tailored to various diseases through target selection, additional effector molecules, and the inclusion of persistence genes. They can be further enhanced through genetic switches to allow selective activation or inactivation of CAR T cell activity as the disease process changes over time.
DMM based therapy
Disease modifying molecules (DMMs) are agents designed to alter the course of diseases by targeting specific pathways or mechanisms involved in disease progression. In the context of neurodegenerative diseases, these molecules play a crucial role in improving therapeutic outcomes by modulating the neural environment, enhancing CAR T cell persistence, and reducing adverse effects. By targeting processes such as inflammation, oxidative stress, and neuronal survival, DMMs help to improve the performance of CAR T cells.
Integrating disease modifying molecules into CAR T cell therapy aims to create more robust and durable responses. These molecules enable CAR T cells to function optimally and adapt to the local microenvironment as physiological changes occur. Consequently, DMMs have the potential to significantly enhance the therapeutic efficacy and safety of CAR T cell therapy, offering new hope for patients with conditions such as Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis (ALS).

